If you are fatigued, get more fatigue
You know what they say to people with hangovers? To have another drink, that is to say to heal evil with evil. Not sure it works for alcohol. But it does for fatigue. Let’s take a look. To begin with, we should remember that before the word fatigue was utilized, we used to talk about neurasthenia. This word has its origins in the Greek words ‘neuron’ (see how easy Greek is) and ‘asthenes’, which literally means ‘weak’. Indeed, muscle atrophy has been described as having a deleterious effect on fatigue (e.g. [1]). This cachexia associated with, for example, certain cancer treatments or advancing age (i.e. sarcopenia) is not the only cause: there may also be what is called a reduction in specific strength. Basically, not only do we have less muscle, but the force produced per unit of muscle mass also decreases. A kind of double punishment. So that’s the potential role of strength. Can we compensate for a relative weakness by a better endurance? In theory yes, but the metabolism is often altered by the disease or by the treatment. Disease can also affect the ability to resist to acute fatigue that occurs during exercise. The theoretical rationale is presented in the figure below.
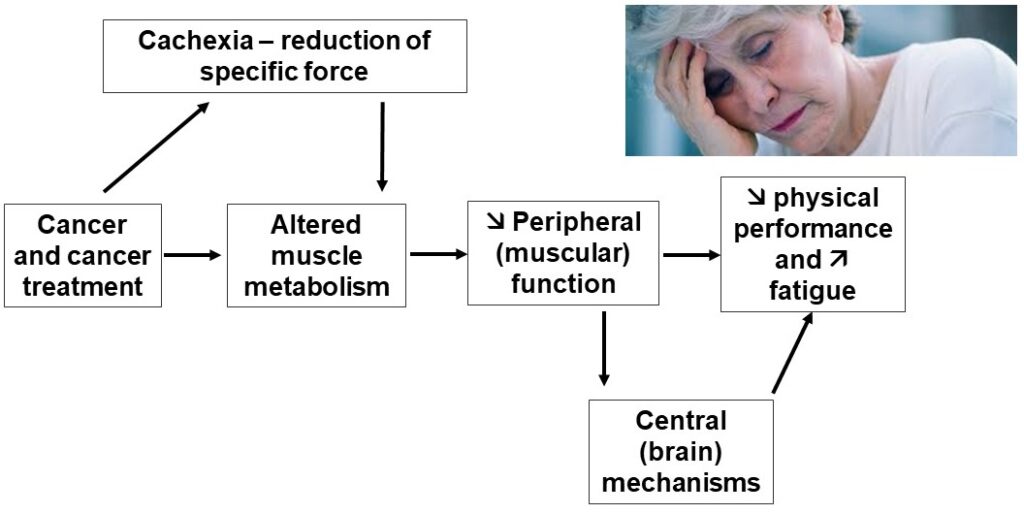
Contrary to what one might think, there is not much experimental evidence of impaired resistance to acute fatigue in patients with chronic fatigue. Moreover, the few studies that have assessed resistance to neuromuscular fatigue in patients have mainly used localized isometric fatiguing exercises. However, very few activities of daily living involve sustained isometric contractions since the Squat toilet is not used anymore. A Danish team has shown that, depending on the ‘ecological’ value of the objective fatigue test, the links with subjective fatigue exists or does not exist [2]. We thus need to think about how we should measure fatigue.
An innovative ergometer
For this reason, we have developed an ergometer (see the picture below) which allows to measure acute fatigue during cycling, i.e. a dynamic exercise involving large muscle masses [3]. This is more similar to daily life when you have to walk, climb stairs, etc. Moreover, with a small system for locking the pedals, which are themselves instrumented, we can measure fatigue straight away, without waiting for the patient to get off the bike and sit in a chair dedicated to measuring force, as it is usually done. We also built a progressive test that allows to mitigate the patient’s motivation [4, 5]. In other words, we no longer wait for him/her to give his maximum to judge his fatigue, but we test the patient at sub-maximal levels of fatigue. Again, this is more similar to daily life.

The results [6] obtained with this original ergometer in patients in remission of cancer show that an attenuated resistance to muscle fatigue on this type of exercise (patients in red on the figure below) can explain in part the subjectively perceived chronic fatigue. As seen in the graph at the top right of the figure below, this difference was apparent during the earliest stages of the progressive test, i.e. exercise intensities corresponding to simple activities of daily living. Climbing stairs, shopping, managing children, cleaning: these are some of the tasks that daily life imposes on us hour after hour, day after day. All of these could lead to an accumulation of fatigue over time [7].
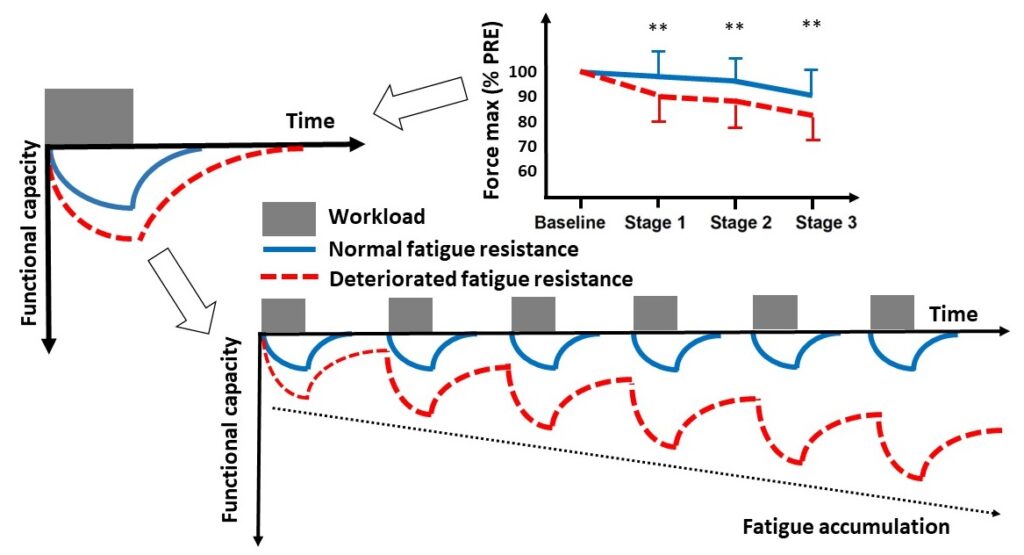
The vicious circle of fatigue
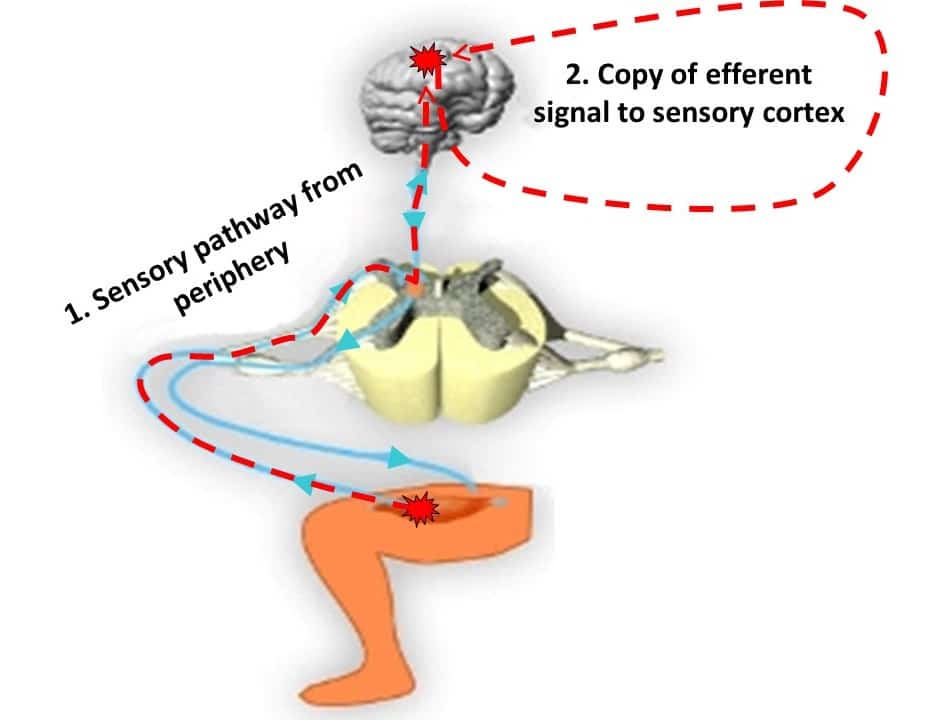
The reality may be different from the daily fatigue accumulation shown in the figure. It is more likely that patients who are less resistant to acute fatigue reach their perceived fatigue ceiling (the moment when they have enough) earlier in the day. As a result, they will be less active in their daily lives. And would therefore enter what is commonly called ‘the vicious circle of fatigue’ [8]: less physical activity leads to deconditioning which in turn leads to more fatigue, which reduces the level of physical activity, etc. How does the brain know that the muscle is more fatigued? By the feedback and feedforward mechanisms described in the figure below. On the one hand, receptors located at the contact of the fibers inform the brain of what is happening below. The Edward Snowden of muscles, so to speak. Or let’s say the Bennet Omalu. On the other hand, to pedal at the same power when our muscles are getting fatigue, the brain has increase central command to the exercising muscles. It has been measured that the electrical signal recorded in the muscles was up to 10% higher in fatigued patients. The brain is capable of feeling this kind of thing. In this study [6], the clinically non-fatigued subjects also had higher cardiorespiratory capacities and lower TNF-a values. The latter induce a syndrome called “sickness behavior” characterized by apathy, anxiety, and loss of appetite, which is similar to depression. We come back to this. In any case, we have some clues to explain the beneficial role of physical exercise. Other studies have shown that fatigue in the elderly is associated with impaired physical function and reduced performance in activities of daily living [9]. In summary, chronic fatigue is not just a matter of deteriorated resistance to muscle fatigue… but the later does play a role.
Hitchcock is not interested
Physical inactivity is correlated with fatigue: more fatigued patients generally report lower levels of physical activity (e.g. [10]). At this point, we have to admit that it’s a chicken and egg situation: do less fatigued patients have more juice to move or does physical activity really have beneficial effects? In order to decide, we need to look at interventional studies in which patients exercise. We are not going to keep the suspense: it works. If we take another look back, we see that this is not entirely new. In an issue of the Journal of Nervous and Mental Disease from the 1890s, it can be read that the ‘bicycle’, a recent invention for the time, allows “measured efforts, varied displacements, continuous changes of scenery”, affecting “happily” the sensibility. The undisputed result: “neurasthenics who used the bicycle were cured much sooner than those who did not use this instrument”. Or again, “walking, slow, prolonged, calculated, which would ensure a minimal muscular engagement” slowly restoring “in neurasthenics the confidence in their own abilities and energy” [11]. In short, even though it may seem counter-intuitive at first sight, physical activity is recommended in the management of fatigue: it has been working for a long time and it even works very well! And that’s good because there are not that many other options. We will not go so far as to say that there is no salvation without exercise, but this is true: in cancer, for example, with a few exceptions, pharmacological treatments have not proved their effectiveness [12] and are not recommended [13]. Conversely, the evidence for the efficacy of physical activity (which could be extended to other non-pharmacological therapies, but the reality is that for fatigue, it is mainly physical exercise) is numerous, both during and after treatment [14]. The first meta-analysis on the topic in cancer was published in… 2005 [15]. Since then, more than 20 meta-analyses gone in the same direction of a benefit of exercise in lowering the level of perceived fatigue [8]. If we really want to look for the smallest detail, we should note that the majority of studies in which physical activity was used to combat fatigue were carried out during treatment, less than 25% being conducted post-treatment. In addition, very few randomized trials of physical activity interventions include fatigue as an eligibility criterion [16]. But that’s all we can argue. In short, physical activity is magic. Now, let’s see why it works.
Why does exercise works to alleviate fatigue?
Go back to part 1 of this paper, in particular the figure that shows the potential causes of fatigue and you’ll have your answer. Basically, physical activity can potentially benefit everything described in this figure. Or almost, because there are one or two exceptions. For example, being active does not increase the number of red blood cells. Exercise still improves oxygen transport by increasing blood volume. And there are so many other positive consequences: exercise improves sleep (e.g. [17]), reduces inflammation [18], I am not even talking about muscle or cardiorespiratory endurance, as this is so obvious. The same goes for depression or anxiety, there are enough studies on the subject. Sometimes physical activity can be beneficial for disease-specific reasons. For example, one study examined the influence of 12 weeks of training on muscle strength (knee extensors and flexors), walking performance, stair climbing ability, self-reported fatigue, mood and quality of life. The researchers found that the intervention reduced the subjective level of fatigue as hoped, but that was not all: the reduction in fatigue was possibly related to an increase in the ability of the nervous system to activate muscles (voluntary activation), a level of activation that they had otherwise found to be deficient in people with multiple sclerosis [19]. In short, the patients are able to recruit more muscle fibers after than before training. To better understand the importance of training, a few other studies have attempted to link changes in fatigue with changes in other parameters measured in randomized controlled trials. Many of these training studies have involved cancer patients. For example, by decreasing oxygen consumption for a given effort and increasing strength at the same time, the relative intensity of activities of daily living is lowered, making the task easier [20]. The same is true when training improves vascular health, which may be compromised by chemotherapy [21]. However, sometimes studies are not as successful as they could be. For example, sometimes there is a decrease in fatigue without any link to the change in physical qualities [22] … or the opposite: an improvement in fitness levels without any effect on fatigue [23]. You cannot win every time, this is true in any research.

A couple of rules
In any case, it is quite logical that the majority of studies have concluded that physical activity is a good way to fight fatigue. Some of these studies are cancer-related: moderate-intensity aerobic training programs lasting at least 12 weeks (three sessions per week) can significantly reduce fatigue during and after treatment [14]. Combined moderate-intensity aerobic and resistance training sessions performed two to three times per week, or moderate-intensity resistance training twice per week, may also be effective [14]. Supervised training appears to be more effective in reducing fatigue than activities performed independently at home [12]. Furthermore, it seems that the reduction in fatigue is mainly observed when physical activity programs are started at the beginning of treatment, although fortunately they can still be effective after treatment [8]. But to a lesser extent. Also, the effects of exercise on fatigue are better for moderate to vigorous vs. low intensity exercise [14]. There is, however, insufficient evidence of a dose-response relationship: beyond 150 minutes of aerobic exercise per week, the reduction in fatigue does not appear to be more significant [14]. Here again, we must remain critical and look for the smallest detail. It is not clear that these interventions are feasible or effective for everyone, especially patients with more severe fatigue. Sometimes fatigue can be a significant barrier to participation in training programs and can even lead to post-exertional malaise [24]. This, sometimes quite high, increase in fatigue after exercise has often been reported in patients with chronic fatigue syndrome. It is even one of the rare contraindications to physical exercise in patients to my knowledge. Some people do not hesitate to speak of an unethical treatment [25]. It is not impossible that patients with long Covid will, at least for some of them, become exercise intolerant even though it is too early to conclude about it.
One size does not fit all
An important and still unanswered question is what type of exercise is best to fight fatigue? Again, in cancer, results of meta-analyses indicate that aerobic exercise interventions are associated with statistically significant reductions in fatigue levels, whereas the effects are more mixed for strength exercises [26]. According to the National Cancer Institute [12], the optimal conditions under which physical activity is likely to reduce cancer-induced fatigue (type of exercise, intensity, duration and frequency, practice environment, etc.) remain debated. The treatment of fatigue is complex and needs to be individualized [27]. This is quite normal given all the above, the complexity, the plurality of causes. It is even impossible for a single approach to training to be optimal. It is the famous ‘one size does not fit all’. Take exercise intensity for example: to improve aerobic fitness, we know that intensity is key. But if training sessions are too difficult, it will only reinforce fatigue in some patients. It is therefore necessary to tailor training to the patient’s capacities, which implies a good evaluation of the intensity domains via thresholds or critical power when possible. In all cases, the fatigue induced by the training sessions should be monitored by a visual analog scale [13] or the ROF presented above [28]. Certain patient characteristics must also be considered. For example, we will not put someone with balance problems on a treadmill. Primum non nocere (first do no harm) is the first principle of caution that medical students learn. The placement of an implantable catheter may also interfere with some weight training exercises. Beyond these very practical but essential considerations, our team has proposed that a targeted intervention on the etiology of fatigue can optimize the effects of the program and decrease fatigue [29]. Some examples are shown in the figure below. In order to target the most beneficial exercises for individuals, it is necessary to consider at least key factors such as age, disease typology (e.g. type of cancer or type of multiple sclerosis), medical co-morbidities (e.g. overweight, diabetes), sports history and, most importantly, personal preferences [20]. The most important thing is that in the end, the best treatment in the world can only work… if you take it. So even if the training is not optimal, in the end it is the patient who decides. Especially for a treatment, physical activity, which must be taken for life. If your thing is walking in the countryside, we won’t insist on making you lift weights in a cold room lit by dim neon lights. The key is to have fun moving.
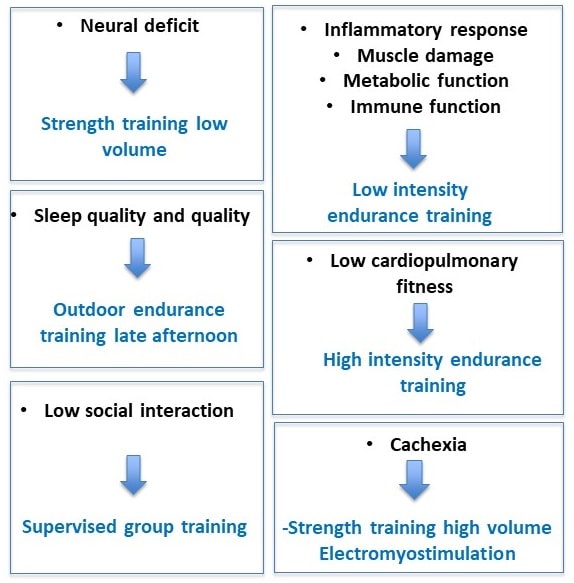
In conclusion, if there is one symptom for which physical activity works miracles, it is fatigue. Drug treatments have not proven their effectiveness and are not recommended, especially in the long term. And you can’t drink 25 espressos a day, even if caffeine is no longer considered a doping product. In cancer for instance, more than 20 meta-analyses have shown the benefits of physical activity, and even if we don’t know precisely why it works, it would be a huge mistake to move. Exercise physiologists and coaches have to work hard to build programs that are tailored to the causes of fatigue. No to individualism, yes to individualization.
References
1. Andrews PLR, M.G., Hickok JT, et al. , Mechanisms and models of fatigue associated with cancer and its treatment: evidence of pre-clinical and clinical studies, in Fatigue in cancer, J. Armes, M. Krishnasamy, and I. Higginson, Editors. 2004, Oxford University Press;: Oxford.
2. Taul-Madsen, L., et al., A Head-to-Head Comparison of an Isometric and a Concentric Fatigability Protocol and the Association With Fatigue and Walking in Persons With Multiple Sclerosis. Neurorehabil Neural Repair, 2020. 34(6): p. 523-532.
3. Doyle-Baker, D., et al., An Innovative Ergometer to Measure Neuromuscular Fatigue Immediately after Cycling. Med Sci Sports Exerc, 2018. 50(2): p. 375-387.
4. Bachasson, D., et al., Quadriceps function assessment using an incremental test and magnetic neurostimulation: a reliability study. J Electromyogr Kinesiol, 2013. 23(3): p. 649-58.
5. Bachasson, D., et al., Assessement of quadriceps strength, endurance and fatigue in FSHD and CMT: benefits and limits of femoral nerve magnetic stimulation. Clin Neurophysiol, 2014. 125(2): p. 396-405.
6. Brownstein, C.G., et al., Physiological and psychosocial correlates of cancer-related fatigue J Cancer Surviv, 2022.
7. Twomey, R., et al., Neuromuscular fatigue during exercise: Methodological considerations, etiology and potential role in chronic fatigue. Neurophysiol Clin, 2017. 47(2): p. 95-110.
8. Fervers, B., Cancers, in Activité physique – Prévention et traitement des maladies chroniques. 2019, Éditions EDP Sciences.
9. Murphy, S.L. and D.M. Smith, Ecological measurement of fatigue and fatigability in older adults with osteoarthritis. J Gerontol A Biol Sci Med Sci, 2010. 65(2): p. 184-9.
10. Berger, A.M., Patterns of fatigue and activity and rest during adjuvant breast cancer chemotherapy. Oncol Nurs Forum, 1998. 25(1): p. 51-62.
11. Vigarello, G., Histoire de la fatigue. 2020: Editions du Seuil.
12. Inca, Bénéfices de l’activité physique pendant et après cancer – Des connaissances aux repères pratiques. 2017.
13. Foucaut, A.-M., et al., fatigue et cancer, in Référentiels inter régionaux en Soins Oncologiques de Support, Association Francophone des Soins Oncologiques de Support, Editor. 2020.
14. Campbell, K.L., et al., Exercise Guidelines for Cancer Survivors: Consensus Statement from International Multidisciplinary Roundtable. Med Sci Sports Exerc, 2019. 51(11): p. 2375-2390.
15. Knols, R., et al., Physical exercise in cancer patients during and after medical treatment: a systematic review of randomized and controlled clinical trials. J Clin Oncol, 2005. 23(16): p. 3830-42.
16. Twomey, R., et al., Physical activity after cancer treatment in people with cancer-related fatigue: A systematic review of randomized trials with fatigue as an eligibility criterion. . SportRχiv, 2020.
17. Flausino, N.H., et al., Physical exercise performed before bedtime improves the sleep pattern of healthy young good sleepers. Psychophysiology, 2012. 49(2): p. 186-92.
18. Meneses-Echavez, J.F., et al., The Effect of Exercise Training on Mediators of Inflammation in Breast Cancer Survivors: A Systematic Review with Meta-analysis. Cancer Epidemiol Biomarkers Prev, 2016. 25(7): p. 1009-17.
19. Dalgas, U., et al., Fatigue, mood and quality of life improve in MS patients after progressive resistance training. Mult Scler, 2010. 16(4): p. 480-90.
20. LaVoy, E.C., C.P. Fagundes, and R. Dantzer, Exercise, inflammation, and fatigue in cancer survivors. Exerc Immunol Rev, 2016. 22: p. 82-93.
21. Hayward, R., et al., Training enhances vascular relaxation after chemotherapy-induced vasoconstriction. Med Sci Sports Exerc, 2004. 36(3): p. 428-34.
22. Puetz, T.W., S.S. Flowers, and P.J. O’Connor, A randomized controlled trial of the effect of aerobic exercise training on feelings of energy and fatigue in sedentary young adults with persistent fatigue. Psychother Psychosom, 2008. 77(3): p. 167-74.
23. Pinto, B.M., et al., Home-based physical activity intervention for colorectal cancer survivors. Psychooncology, 2013. 22(1): p. 54-64.
24. Twomey, R., et al., Post-exertional Malaise in People With Chronic Cancer-Related Fatigue. J Pain Symptom Manage, 2020. 60(2): p. 407-416.
25. Twisk, F.N. and M. Maes, A review on cognitive behavorial therapy (CBT) and graded exercise therapy (GET) in myalgic encephalomyelitis (ME) / chronic fatigue syndrome (CFS): CBT/GET is not only ineffective and not evidence-based, but also potentially harmful for many patients with ME/CFS. Neuro Endocrinol Lett, 2009. 30(3): p. 284-99.
26. Bower, J.E., Cancer-related fatigue–mechanisms, risk factors, and treatments. Nat Rev Clin Oncol, 2014. 11(10): p. 597-609.
27. Veauthier, C., et al., The Berlin Treatment Algorithm: recommendations for tailored innovative therapeutic strategies for multiple sclerosis-related fatigue. EPMA J, 2016. 7: p. 25.
28. Brownstein, C.G., et al., French Translation and Validation of the Rating-of-Fatigue Scale. Sports Med Open, 2021. 7(1): p. 25.
29. Twomey, R., et al., Tailored exercise interventions to reduce fatigue in cancer survivors: study protocol of a randomized controlled trial. BMC Cancer, 2018. 18(1): p. 757.
